By M. Marin
READ THE FULL XPHYF RESEARCH REPORT
Focused on next-generation diagnostics, drug delivery, and drug development opportunities …
XPhyto Therapeutics (OTC:XPHYF), a bioscience accelerator focused on opportunities in the areas of next-generation diagnostics, drug delivery, and drug development, recently completed its acquisition of 3a-Diagnostics and believes the transaction will accelerate its growth in the point-of-care biosensor market and facilitate the commercialization of certain products in 3a's development pipeline that are in more advanced stages of development.
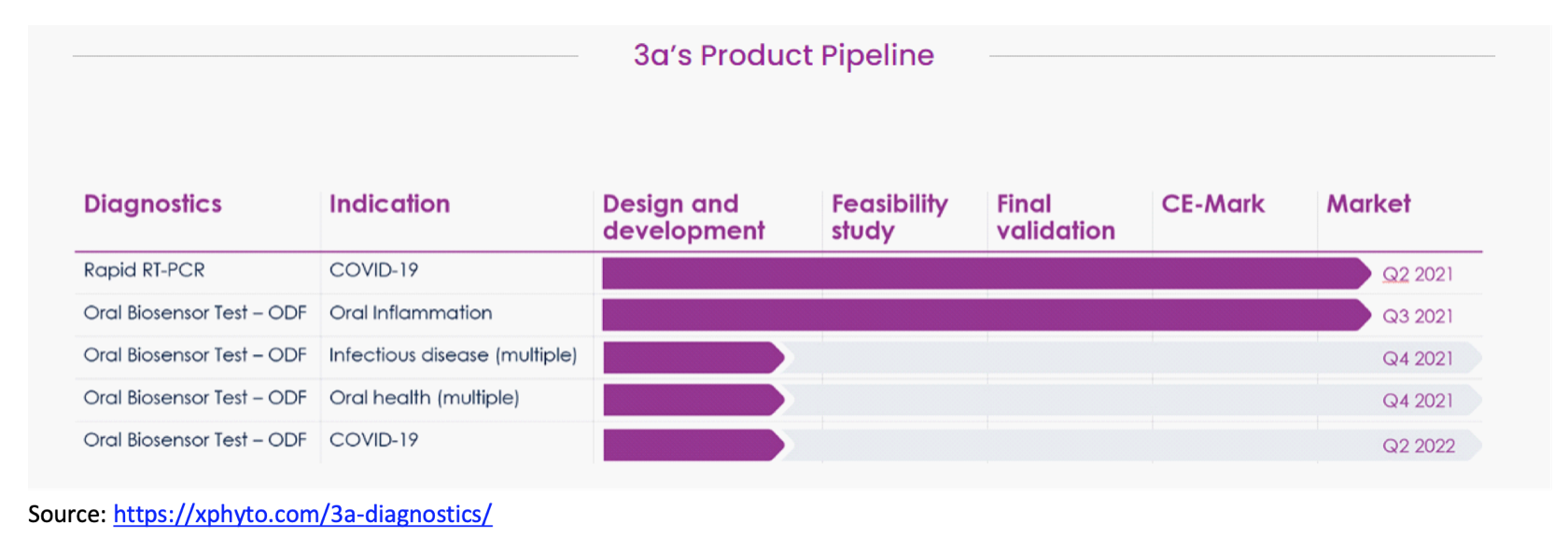
Xphyto is leveraging 3a-diagnostic's expertise and technology to develop and launch oral diagnostic products, beginning with a COVID-19 rapid test. 3a's biosensors are enzyme-activated and developed to be rapid, economical and easy to use for oral screening to detect infectious diseases, including COVID-19, at home or other venues, including point-of-care and high-traffic locations where rapid screening is critical, such as airports and railway stations. XPhyto intends to integrate its thin film technology with 3a's biosensor technology; management believes the combination of these technologies position the company for strong growth and market share gains within the biosensor market.
Rapid point-of-care COVID-19 PCR system launched in Europe
The company is developing a pipeline of low-cost biosensor screening tests and recently launched a rapid point-of-care COVID-19 PCR system in Europe. Importantly, PCR tests are more sensitive and reliable than antigen tests and can detect the presence of coronavirus at earlier stages when the coronavirus material is still replicating within the person's body and the relative viral load is lower. This means that people who are pre-symptomatic and/or asymptomatic but still have been exposed to the virus can test to determine the presence of coronavirus protein in their system. Similar to an antigen test, the sample is collected using a nasal or throat swab. The company notes that PCR tests are the highest standard of COVID testing that have been adopted. XPhyto's RT-PCR test is rapid, low-cost and disposable. Importantly, it requires no additional equipment other than a small portable proprietary Covid-ID lab and off-the-shelf PCR device, so the test can be conducted almost anywhere.
With COVID-19 cases spiking in many markets, the need to test for a variety of reasons, such as ahead of travel or to attend school in-person continues to grow and is expected to persist for the foreseeable future.
Transdermal patches, oral dissolvable drug (ODF) formulations & neurological products leveraging cannabinoid and psychedelic compounds
In addition, the company is also developing precision transdermal (TDS) patches and oral dissolvable drug (ODF) formulations, as well as neurological products leveraging cannabinoid and psychedelic compounds, among other therapeutic products. XPHYF intends to leverage its transdermal and sublingual delivery technology to introduce therapeutics for a variety of conditions. Xphyto's Vektor subsidiary focuses on developing generic and hybrid-generic drug formulations for neurological conditions through its transdermal and oral dissolvable drug delivery platforms. Products in Vektor's development pipeline target large and growing neurological markets. The company also intends to leverage the cannabinoid and psychedelic expertise and relationships that it has formed to develop and launch innovative medicinal programs.
XPhyto's product pipeline is focused on high-margin products designed to capitalize on growth in both the rapid screening test market and overall thin film drug delivery market, and the rapid growth of cannabis-based therapeutics. In addition to its diagnostic pipeline, the company has an ODF development program underway for the delivery of the active pharmaceutical ingredient cannabidiol to treat epilepsy and an ODF development program for the delivery of the active pharmaceutical ingredient THC to treat several conditions.
The company expects to launch an epilepsy clinical trial in 1Q22. XPhyto expects a European human CBD bioavailability study of treating certain epilepsy using its fast-dissolving CBD oral strips will begin in January 2022. Xphyto believes the study will support that its strips provide efficient and precise dosing and enable the company to move forward with the commercialization of the treatment for patients with certain types of epilepsy. According to management, the FDA and European Medicines Agency have approved other CBD-based medical products for the treatment of certain severe childhood forms of epilepsy.
Enhanced financial flexibility to support growth
The company has improved its financial flexibility to support growth with recent capital issuances. Last month, XPhyto closed a private placement and a convertible debenture unit offering, raising combined gross aggregate proceeds of $7.0 million to support its growth strategy and development efforts, including funding the recent 3a-diagnostics transaction.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives quarterly payments totaling a maximum fee of up to $40,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.