
The physician heading a Phase III clinical trial in Pakistan for a Chinese COVID-19 vaccine candidate has urged people to volunteer for the trial, overcoming the resistance in the country to immunisation programmes.
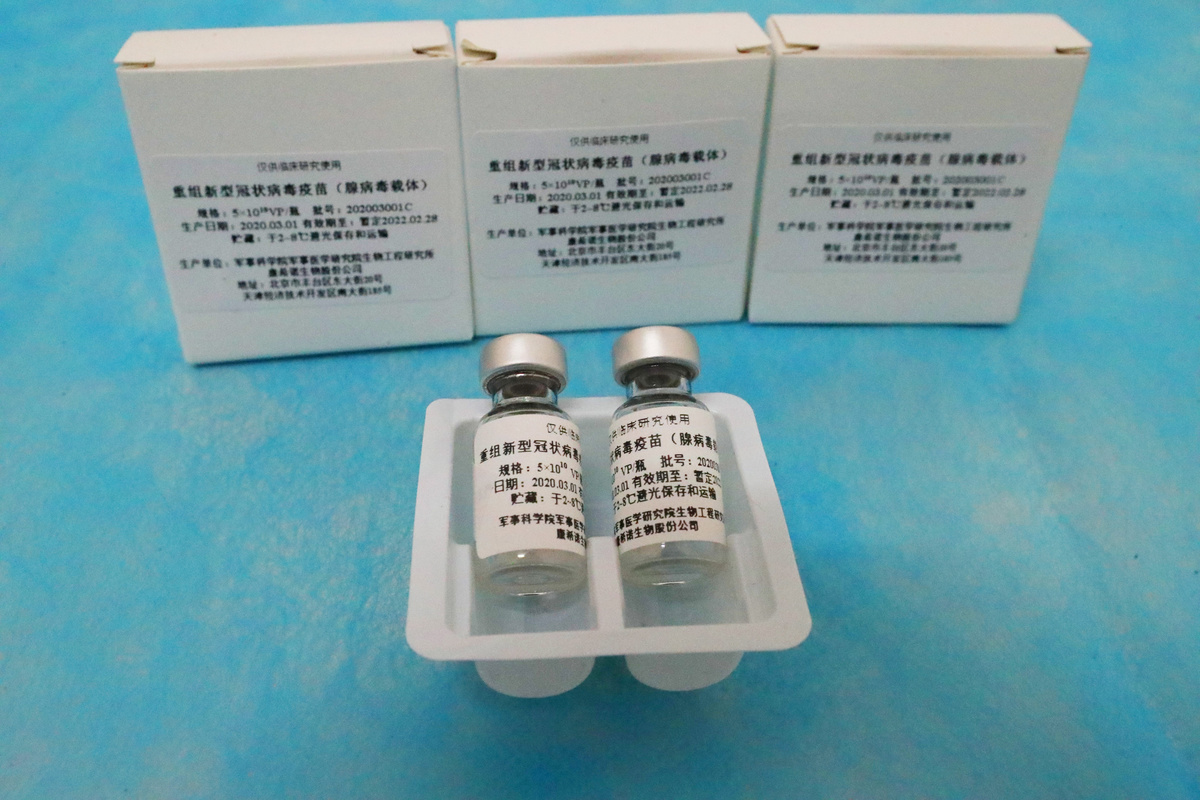
Pakistan launched the trial last week for Ad5-nCoV, a vaccine candidate co-developed by CanSino Biologics and a Chinese military-backed research unit.
It is the first-ever large scale trial in Pakistan, which has grappled with disinformation around other long-established vaccines, and attacks on health workers administering them.
Efforts to eradicate polio, for instance, have for years been undermined by opposition from some Islamists, who say immunisation is a foreign ploy to sterilize Muslim children or a cover for Western spies.
“There are lots of challenges whenever you introduce something new and a vaccine is part of it. Vaccine hesitancy, unfortunately, with a country like Pakistan is also pretty much high,” Ejaz A. Khan, who is heading the trial at Islamabad's Shifa International Hospital, told Reuters on Tuesday.
"People should come and volunteer, people should not be hesitant. They can take part and become part of the team which is fighting COVID-19."
Khan, who has taken part in immunisation drives for three decades in Pakistan, said even existing vaccines had side effects, and hoped Ad5-nCoV would not fall prey to this discussion.
Shifa International, the first of five trial sites in Pakistan, has repurposed a building previously used for COVID-19 testing for the trial, which it hopes will have 2,000 participants.
Volunteers arrive by appointment, and are recruited through NGOs, hospitals, and corporations.
Volunteers must be over 18, not have tested positive for COVID-19, not have immune deficiencies, and not be pregnant for the duration of the trial. A one-time 2,000 Pakistani rupees ($12) compensation for travel and food expenses is provided, Khan said.
The trial's end point, Khan said, is flexible, but one goal is to show the vaccine is 50% more effective than a placebo.

Once proven, Khan said it was expected Pakistan would be provided with several million doses on a priority basis by CanSinoBio.
Pakistan’s National Institute of Health, which is overseeing the trial, did not respond to a Reuters request for comment.
Pakistan reported 541 new cases on Tuesday - taking the total to 312,263 with 6,479 deaths.
($1 = 165.5000 Pakistani rupees)
(Reporting by Umar Farooq; Editing by Raju Gopalakrishnan)








