The nation's first clinical trial to transplant embryonic stem (ES) cells into a patient began at the National Center for Child Health and Development (NCCHD) in Tokyo on April 30.
ES cells, derived from early-stage embryos, are a type of pluripotent stem cells that can differentiate into all cell types. The trial will be conducted on babies with a serious liver disease, and the first transplant of ES cells will be conducted as early as this autumn.
However, Japan faces many challenges in the expansion of regenerative medicine (see below), even though the field includes the use of induced pluripotent stem (iPS) cells, an area of research in which Japan takes the lead.
Transplanting into a baby
"We are finally arriving at the starting line for the development of therapies using ES cells," said Mureo Kasahara, director of NCCHD's Organ Transplantation Center.
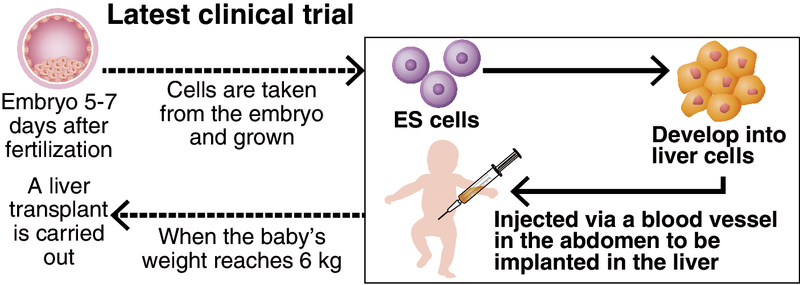
The trial will be conducted on babies with hyperammonemia, who are born without the ability to make a liver enzyme that degrades harmful ammonia. About 10 babies are found to have this intractable disease each year in Japan. The only existing treatment for it is a liver transplant. However, the transplant cannot be conducted on babies weighing less than 6 kilograms. There have been cases in which the baby dies before his or her weight reaches that level.
ES cells are created from early-stage embryos that were created for fertility treatments and would otherwise be discarded. The NCCHD created ES cells for the clinical trial with the consent of a couple who received such fertility treatment.
In the clinical trial, normal liver cells cultured from ES cells will be injected into a baby in such a way that they become implanted in the baby's liver. Until the baby grows enough to receive a liver transplant, treatment is provided to prevent the deterioration of the baby's liver condition.
It will be the first time in the world for liver cells created from ES cells to be transplanted into a patient. However, there have been cases using healthy leftover cells after a liver transplant, in which the patient's condition improved. The effectiveness was also confirmed in experiments on animals.
The NCCHD will conduct a number of checks to confirm the processes of the clinical trial. Then, they plan to transplant such liver cells into a total of five patients over a two-year period starting this autumn to confirm the safety and effectiveness of the therapy.
Akinari Fukuda, a doctor who is in charge of the clinical trial, said: "By using ES cells, we can prepare liver cells when a liver transplant is necessary. We would like to establish a therapy to serve as a bridge until a liver transplant is conducted."
Japan lags other countries
Human ES cells were created for the first time in the United States in 1998. Due to ethical concerns over the use of fertilized eggs, government funding for ES cell-related research was temporarily cut off in the United States. However, the research has been continued mainly by companies, and today many clinical trials for the application of ES cells to treat spinal cord injury, diabetes and intractable eye diseases are under way in the United States, Europe, China and South Korea.
Yoji Sato, a division chief at the National Institute of Health Sciences and an expert in regenerative medicine, said, "There have been no reports that ES cells became cancerous or caused other serious symptoms, and the world's first drug using ES cells may be launched in the United States next year."
On the other hand, partly due to ethical concerns, the creation and use of human ES cells had been limited to those intended to be used for basic research until 2014 in Japan. Since the emergence in 2006 of iPS cells, which are created from somatic cells rather than embryonic cells, the government's budgets have concentrated on research into iPS cells. Also in clinical research, iPS cells took the lead.
A university professor who has been involved in regenerative medicine said, "Research on ES cells has been put aside, resulting in Japan lagging behind the United States in the field."
While Japan is now behind other countries in ES cell research, Norio Nakatsuji, a professor emeritus at Kyoto University who specializes in stem cell biology, said: "ES cells are highly stable in terms of their quality. Knowledge that has been accumulated so far can be used for iPS research, too."
Kyoto University and the NCCHD are expected to promote full-scale creation and distribution of ES cells for clinical use this fiscal year. Research on this field likely will be promoted also at universities and research institutions in Japan.
"In Japan, as in other countries, it is important to conduct research both on ES cells and iPS cells," Nakatsuji said.
-- Regenerative medicine
Using ES cells, iPS cells and other stem cells that change into specific cells such as blood cells, can make it possible to regenerate or restore the functions of organs and tissues that have been damaged due to injury or disease. It is expected that this medicine will help such things as improvement in paralysis symptoms caused by spinal cord injuries and treatment of intractable eye or nerve diseases.
Read more from The Japan News at https://japannews.yomiuri.co.jp/







