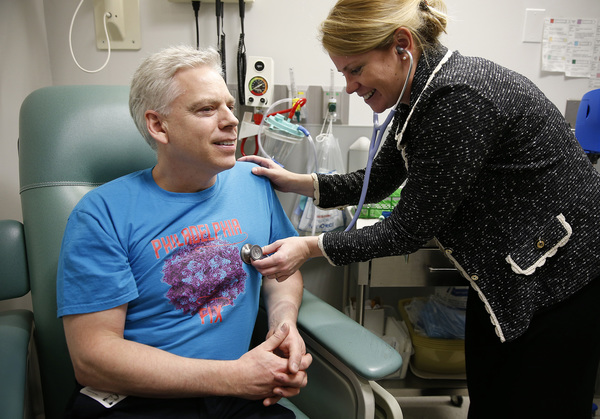
At a Home Depot not long ago, Jay Konduros was hefting a 40-pound box of shelving into his cart when it suddenly slipped _ and bam!
The blood welled up from a gash in his shin, and his mind flashed back to a lifetime of caution and pain.
That minor mishap with a snow-shovel that put him out of commission for weeks, his leg so swollen he could not see his knee.
The many days he told friends he could not come out to play, but didn't want to say why. The times his mother rushed him to the hospital, carrying him because he couldn't walk.
But then, staring at his gashed shin at Home Depot, he thought this time might be different.
Konduros was born with hemophilia, the blood clotting disorder.
Any time he banged up an arm or leg, and sometimes even without any apparent injury, he might develop the swelling and pain that are the hallmarks of dangerous internal bleeding. Each time it happened, he had to infuse himself with costly synthetic clotting factor _ a lifetime tab that so far had run into the millions.
Then last year, the 52-year-old was among the first in the world to receive a one-time, experimental treatment: a dose of genetically engineered particles that would enable his liver to make enough clotting factor on its own.
If he was lucky, this one-shot treatment _ developed by Spark Therapeutics, a biotech startup spun off from Children's Hospital of Philadelphia _ would ward off his disease for what could be many years.
The Home Depot accident, near his home outside Toronto, represented an unplanned test of the treatment. In the hours and days after the heavy box slammed into his leg, Konduros waited and watched.
There was no bruising. No swelling. No debilitating pain.
"You kind of smile and think, 'Ok, that's abnormal,' " he said.
.jpg?w=600)






