PITTSBURGH _ Generic drug giant Mylan reported lower than expected profits in the second quarter and cut its earnings guidance for the full year because of delayed launches of two major drugs and bigger than forecast declines in EpiPen sales.
Mylan shares rose 29 cents, or 1 percent, Wednesday to close at $32.08
Since hitting a peak for the year of $45.28 in early March, the company's shares have slumped 30 percent. Investors have been concerned about delays in regulatory approvals for generic versions of big-name drugs Copaxone and Advair, and price erosion in the generic drug industry.
Mylan said Wednesday it earned $297 million, or 55 cents per share, for the second quarter, up strongly from $168.4 million, or 33 cents, in the year-ago quarter.
But excluding acquisition-related costs and other special items, adjusted earnings were flat at $589.9 million vs. $592.4 million in 2016, while per-share profits fell 5 percent to $1.10 from $1.16. That missed analysts' consensus estimate of $1.16.
Results were released before the stock market opened on Wednesday.
Revenue was $2.96 billion, up 16 percent from $2.56 billion in the same quarter last year. Sales in the North America segment fell 9 percent.
Mylan lowered its adjusted earnings outlook for the full year to between $4.30 and $4.70 per share, down from $5.15 to $5.55, citing "the (U.S.) region's ongoing challenges and the uncertain U.S. regulatory environment" for product approvals. It also lowered its target for 2018 from $6 a share to "at least" $5.40.
"Yes, this was a tough quarter and it will be a challenging year," CEO Heather Bresch told analysts in a conference call. She added that the company was being managed for the long term.
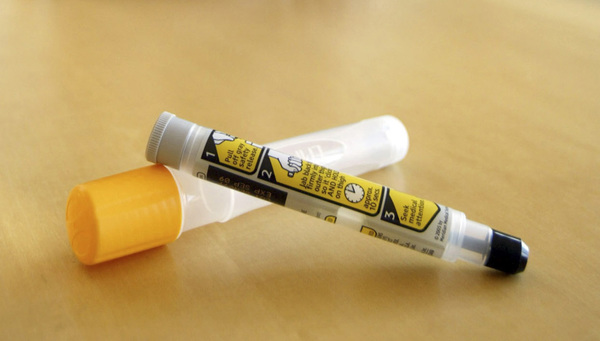
The company said sales of its EpiPen emergency allergy shot fell more than expected in the wake of increased competition and the release in December of an authorized generic. Mylan started selling the generic to compete with its own brand name product amid public outcry and congressional scrutiny over the spiraling cost of the EpiPen.
Last year, EpiPen controlled some 95 percent of the auto-injector market. Mylan's marketshare has since eroded to around 70 percent, including sales of the EpiPen and the authorized generic, the company told analysts in a conference call Wednesday.
Besides industry concerns and outrage over the EpiPen, Mylan faced some negative publicity in June when shareholders overwhelmingly rejected the company's executive pay practices, showing a growing dissatisfaction over multi-million-dollar compensation packages that included nearly $100 million for chairman Robert Coury in 2016.







