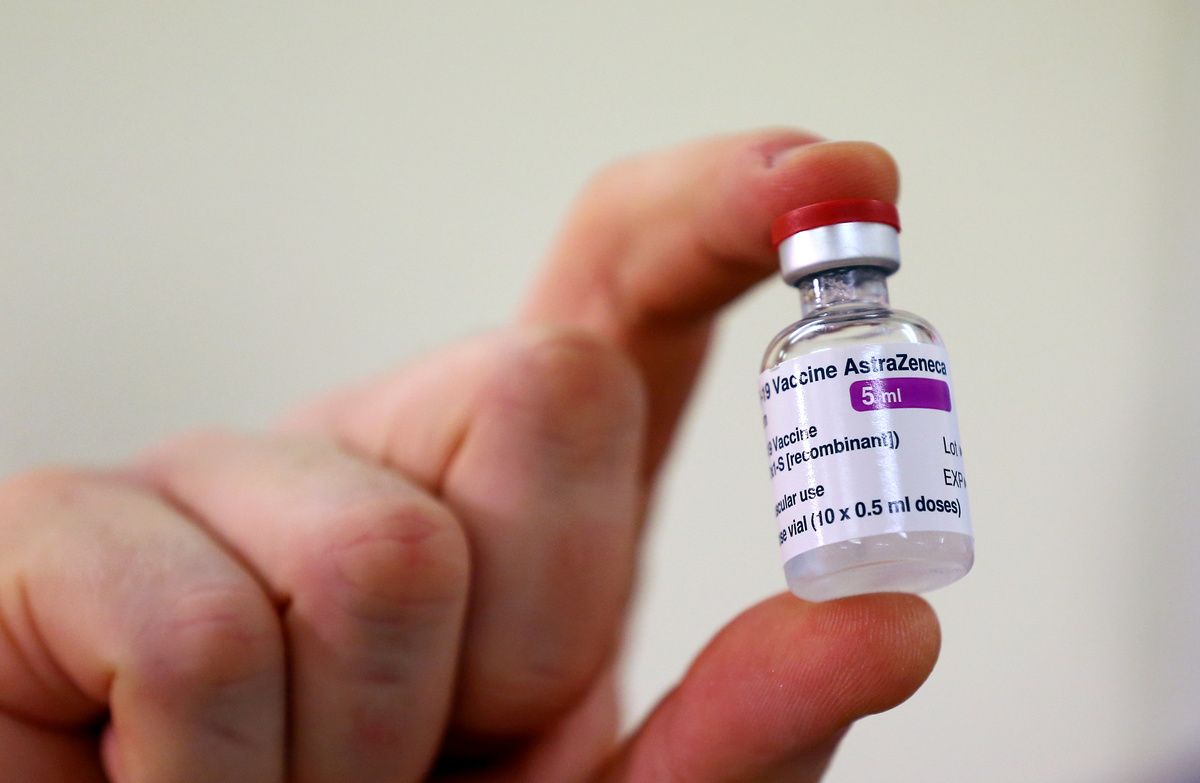
Brazil's health regulator Anvisa said late on Saturday it had approved the import of 2 million doses of the COVID-19 vaccine developed by AstraZeneca and the University of Oxford, although the jab is not yet approved for use in the country.
Anvisa said it approved the importation request from federal government-affiliated biomedical center Fiocruz on Thursday.
Importing the doses prior to approval will enable vaccination to begin as soon as Anvisa okays its use, the regulator said. Brazil has yet to approve any vaccine.
Fiocruz will apply for emergency use of the vaccine by Wednesday, its president, Nísia Trindade, said last week.
Brazil has recorded the second-deadliest outbreak of COVID-19 after only the United States. South America's largest country has the third highest tally of coronavirus cases, with the health ministry registering more than 7.7 million, after the United States and India.
(Reporting by Jake Spring; Editing by Daniel Wallis)







