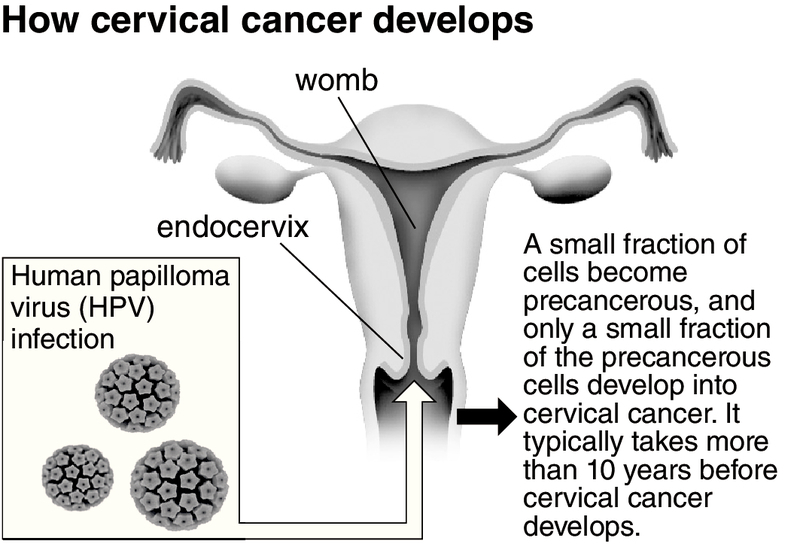
It has been five years since the Health, Labor and Welfare Ministry stopped actively recommending the human papilloma virus (HPV) vaccine (see below), which protects against a virus that can cause cervical cancer. The ministry had been recommending it by informing eligible individuals about routine vaccinations in writing, but stopped doing so because several reports suggested the vaccine can cause negative side effects.
While those in favor of recommending the vaccine and those against it remain at loggerheads, the number of people choosing to be vaccinated is on the decline. It is essential to reexamine this vaccine in light of new data that has emerged during this five-year period.

Less than 1% get vaccine
"None of my classmates knew about this vaccine," said a 16-year-old high school student from Gunma Prefecture who had her third administration of the HPV vaccine in March.
Girls aged 12 to 16 are eligible for the routine vaccination against HPV, which is administered in three sessions. At one time, it was estimated that more than 80 percent of those eligible had been vaccinated, but that figure has fallen to less than 1 percent today.
The girl got the vaccination at the advice of Yuichi Sato, an obstetrician and gynecologist at a hospital in Takasaki, Gunma Prefecture. When the student's mother, 47, asked for the doctor's opinion on the vaccine in July last year, he suggested bringing her daughter in for it.
In early August, he spoke to the mother and daughter about both the benefits and side effects of the vaccine. The daughter was convinced, saying, "I only had a scary image of the vaccine before, but I came to understand its effectiveness," and so she decided to get the vaccine. She and her two siblings were born at that particular hospital, which reinforced her decision with a sense of trust in the institution.
Lawsuits over side effects
In November 2010, the government began to subsidize the cost of the HPV vaccine. In April 2013, it was reported that a number of young girls started suffering from intense pain and movement disorders after the routine vaccination system began.
On June 14, 2013, the ministry decided to stop recommending the vaccine until it could provide more information in response to citizens' concerns. After the information on routine vaccination stopped being delivered to eligible individuals, the vaccination rate sharply declined.
Even so, the routine vaccination is still generally offered for free to girls of the eligible age.
Tasked with looking into the cause of symptoms that emerged post-vaccination, a panel of experts at the ministry examined global clinical data and concluded in January 2014 that it was a physical response to pain and anxiety at the time of vaccination rather than to any of the ingredients of the vaccine itself.
The ministry also assembled a research team to establish treatment for the symptoms, and set up treatment centers at 90 cooperating medical institutions across the nation. In January this year, the ministry revised the explanatory pamphlet on the vaccine's efficacy and side effects, updating it with the latest information.
However, 123 women who have not recovered from serious symptoms are seeking damages from the government and two pharmaceutical companies for the harmful side effects they allege were caused by the ingredients of the vaccine, and court cases are ongoing in four district courts across the country.
Representative plaintiff Nanami Sakai was left with a serious learning disorder and movement disorder after vaccination. A 23-year-old university student from Fujimino, Saitama Prefecture, she now uses a wheelchair.
Though she visited 25 medical institutions seeking help, she couldn't get a satsifactory response. She even experienced harsh words from some doctors, being told it must surely be hysteria, and that she should get used to life in a wheelchair.
Now she visits Kagoshima University Hospital, about 960 kilometers away from her house. Nanami's father, Hideo, 59, said, "There are very few medical institutions, including cooperating medical institutions, that are able to respond to the kind of neurological symptoms my daughter has."
Detailed explanations
Routine vaccination against HPV is carried out in more than 70 countries worldwide. The World Health Organization is critical of the current situation in Japan, declaring that not using the vaccine will bring real harm.
The Japan Society of Obstetrics and Gynecology and others have called for resuming efforts to actively recommend the vaccine as soon as possible. But the affected women seeking damages and some of the doctors treating them are opposed, so discussions remain deadlocked.
The ministry is reluctant to resume recommending the vaccine in the near future. It seeks to observe how people respond to the revised information pamphlet and explore the most appropriate ways of providing information to citizens, according to the ministry.
Regarding the current situation, Director Nobuhiko Okabe of Kawasaki City Institute for Public Health, who is well-versed on the topic of vaccines, believes that there have been sufficient grounds for resuming recommendation of the vaccine at any time in the past five years.
While calling for resumption of the recommendation, he also says that post-vaccination symptoms should be taken seriously and their causes should be determined. He also insists that, returning to a standard that establishes a relationship of trust between doctors, girls and their families, the vaccination should be implemented only when thoroughly informed consent has been given.
Michiko Yamamoto, a visiting professor at Kumamoto University who specializes in communicating the risks of medicines, believes that it is essential to have in place a process to explain both what is known and what is unknown about the effectiveness and risks of any new vaccine. She asserts that, in the case of the HPV vaccine, the merits were emphasized above all else, and the introduction was hasty. Moreover, if recommendation of the vaccine were to resume, Yamamoto considers it imperative to provide citizens with the most appropriate advice, and carry out follow-up surveys with the people who receive the vaccination in the same way as is done with new anticancer treatments and other medicines.
Gather data and clarify symptoms
In these past five years, there has been an increase in the number of research papers regarding the efficacy of the HPV vaccine.
One notable example is a report that was published in May by the Cochrane Review Groups based in Britain, which aim to promote health care grounded on scientific principles.
After evaluating the results of 26 clinical trials around the world, they found that administering the vaccine reduced the likelihood of developing precancerous lesions from a maximum of 164 people per 10,000 to just two people, which is less than one 80th.
Regarding the question of its effectiveness in preventing cancer itself, a research group from Finland announced at the end of last year that the results from a follow-up survey of about 27,000 women showed that cancer had decreased in the vaccinated portion of the population.
The number of reports that suspect negative side effects caused by the HPV vaccine in Japan make up a mere 0.09 percent of the literature.
Yet, Professor Emeritus Yoshiyuki Kuroiwa of Yokohama City University, whose research is based on the hypothesis that the symptoms that emerge post-vaccination are neurological disorders caused by ingredients in the vaccine, said: "Even if the number is small, it is a reality that there are people who are suffering. We need to ascertain the kinds of people who are likely to be affected."
Lawsuits concerning post-vaccination symptoms are not limited to Japan either, there are also court cases concerning the harmful effects of the vaccine under way in Colombia.
In April of this year, associations of patients from five countries who are suing due to adverse reactions to the vaccine made a joint declaration seeking the development of treatments and support for the affected women.
(From The Yomiuri Shimbun June 7, 2018)
-- HPV vaccine
The two kinds of this vaccine that are available in Japan are Cervarix and Gardasil. Human papilloma virus (HPV), which is spread through sexual intercourse, is a cause of cervical cancer, and the vaccine prevents its infection. About 3.4 million people in Japan have been vaccinated. Around 3,000 of those people have reported symptoms that are suspected to be side effects of the vaccine.
Read more from The Japan News at https://japannews.yomiuri.co.jp/







