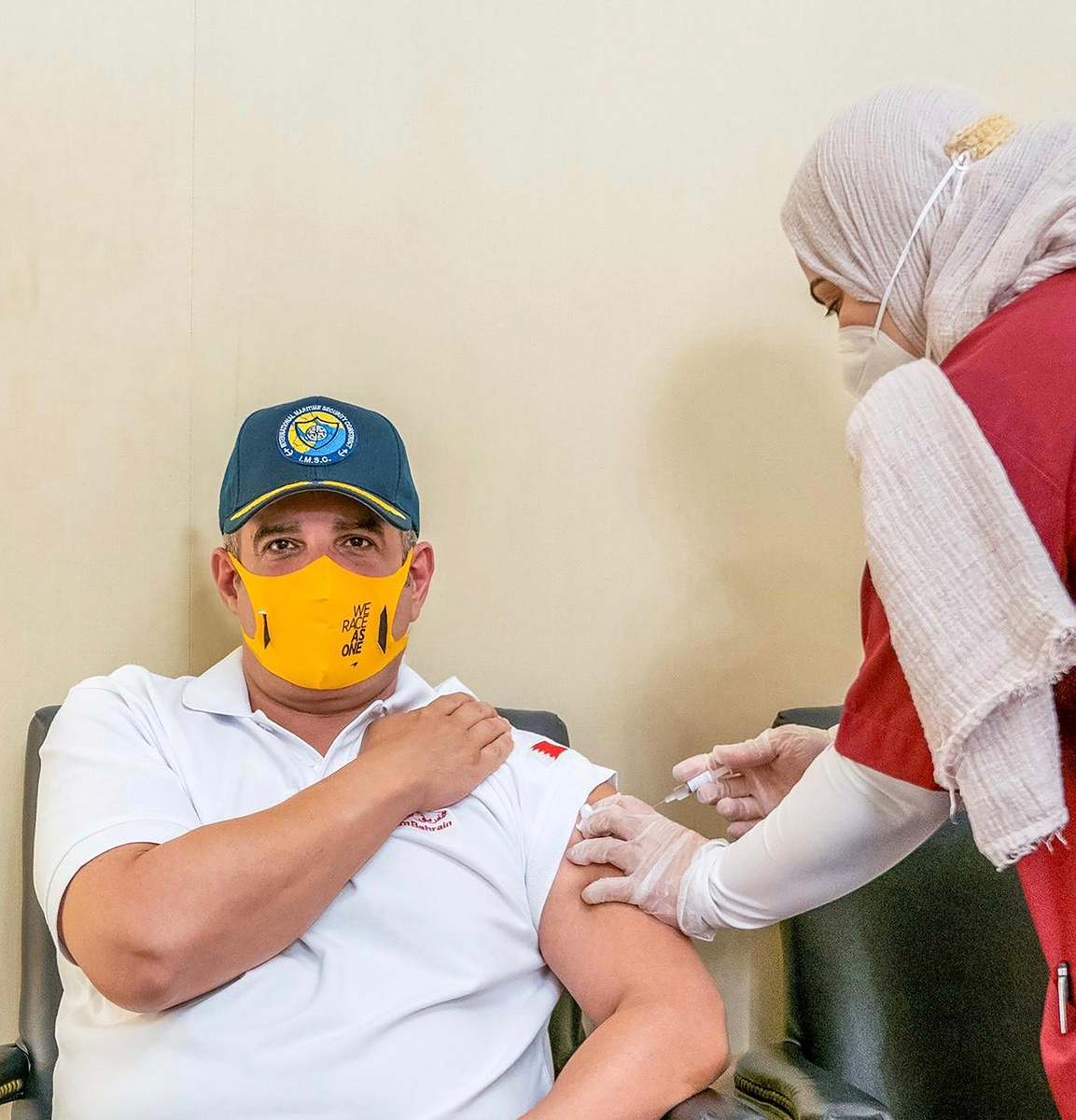
Bahrain said on Sunday it had approved a COVID-19 vaccine developed by China National Pharmaceutical Group (Sinopharm) and launched online registration for the vaccine for citizens and residents.
A statement by the National Health Regulatory Authority did not specify which vaccine of two being developed by Sinopharm had been given the go-ahead, but cited data from Phase III clinical trials that showed an 86% efficacy rate and said Bahrain had participated.
Bahrain's Ministry of Health said in a statement and on its Instagram account that citizens and residents above 18 years of age could register online to receive the vaccine for free.
The data cited was the same as announced earlier this month by the United Arab Emirates from an interim analysis of late-stage clinical trials for an inactivated vaccine developed by the Beijing Institute of Biological Product, a unit of Sinopharm's China National Biotec Group (CNBG).
In July, the UAE started Phase III clinical trials for the vaccine and the trial was expanded to Bahrain, Jordan and Egypt.
Neither CNBG nor Sinopharm was available for comment.
The Bahrain statement said the kingdom had participated in Phase III trials of the approved vaccine and had previously authorised it for emergency use to frontline professionals.
Bahrain earlier this month granted emergency use authorisation for the Pfizer/BioNTech COVID-19 vaccine.
In fellow Gulf Arab state Kuwait, the health ministry on Sunday granted emergency use authorisation for the Pfizer/BioNTech vaccine, state news agency KUNA said.
(Reporting by Hadeel Al Sayegh; Additional reporting by Roxanne Liu; Writing by Ghaida Ghantous; Editing by Gareth Jones and Barbara Lewis)